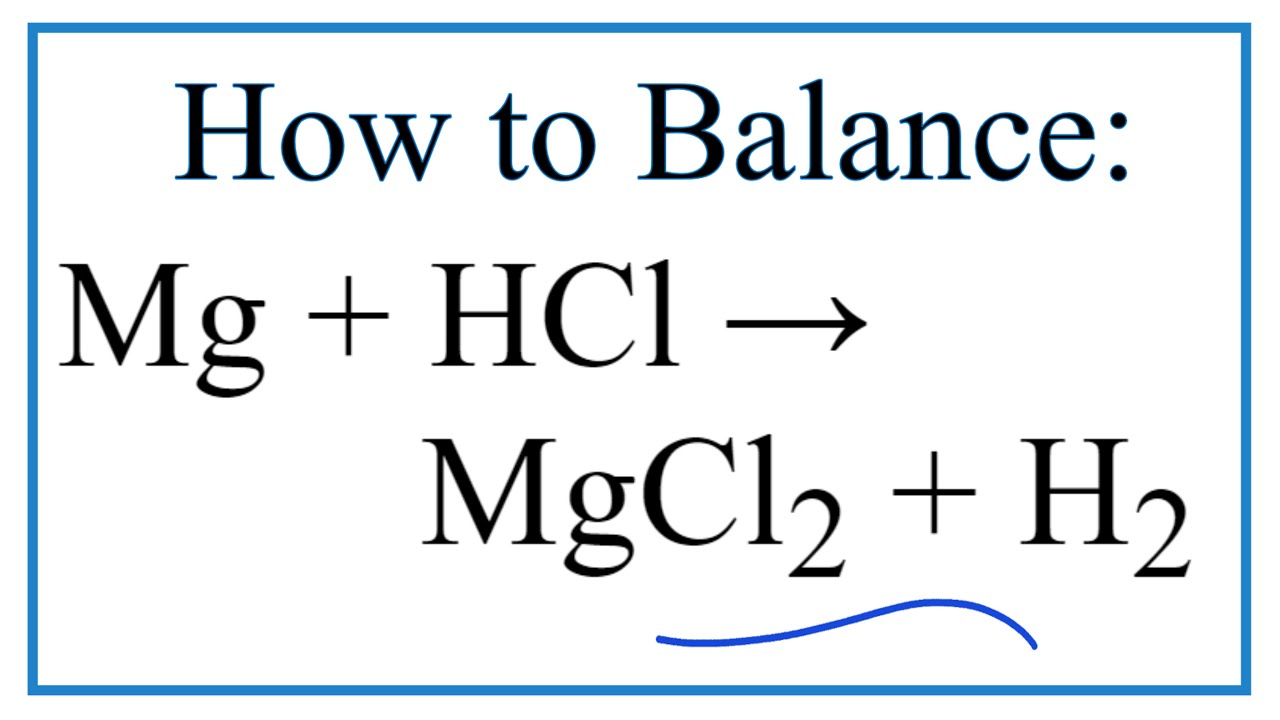
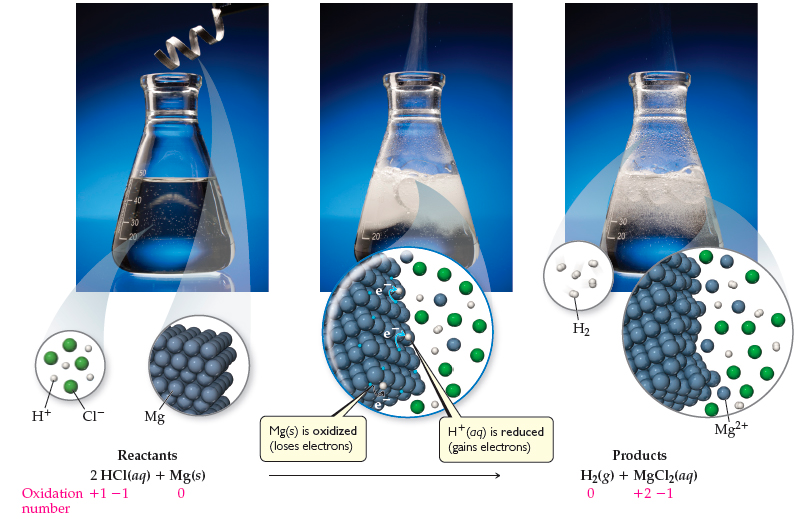
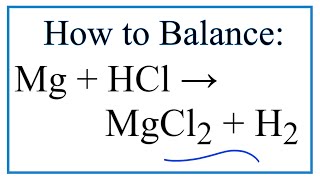
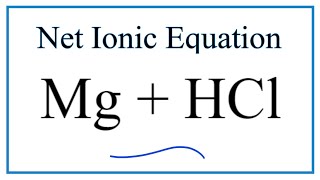Question Video: Identifying the Name of the Gas Produced When Magnesium Metal Reacts with Hydrochloric Acid | Nagwa

One gram of an alloy of aluminium and magnesium when heated with excess of dilute HCl forms magnesium chloride, aluminium chloride and hydrogen. The evolved hydrogen collected over mercury at 0^oC has

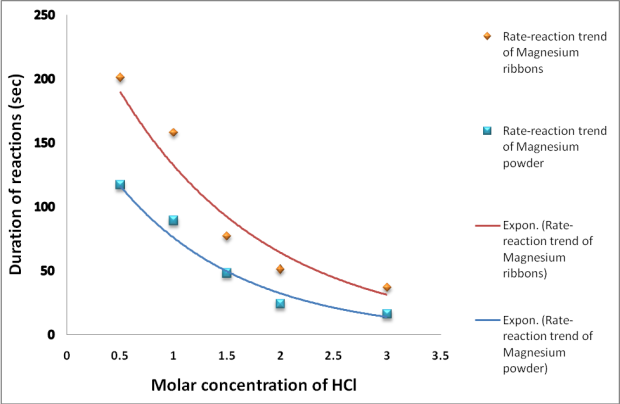
Question Video: Recognizing Why Warm Hydrochloric Acid Has a Faster Rate of Reaction with Magnesium than Cold Hydrochloric Acid | Nagwa